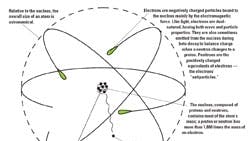
Brushing Up: Structure of the atom
Four hundred years before the birth of Christ, the renowned Greek philosopher, Democritus, suggested that all matter is composed of tiny, discrete, indivisible particles called “atoms.” Although his ideas were rejected for 2,000 years, an English schoolteacher named John Dalton finally saw the light in 1803, developing his atomic theory that forever changed science.
Since that time, physicists have been probing into the structure of the atom, constantly discovering more particles — and particles within particles — many of which exist for a split second and decay all but instantly. There are literally hundreds of such subatomic particles, some of which are virtually without mass, existing only as points of energy or charge in space.
Despite new findings, however, the basic structural components of the atom are well established, and can be broken down into what physicists believe are the most rudimentary parts.