Heart disease accounts for nearly half of all deaths in the developed world. Although heart transplants are still the most effective medical intervention for the disease, the demand for donor organs far exceeds the supply. To address this imbalance, researchers are exploring the potential use of mechanical heart assistance devices. One such device under development at the University of Leeds in the UK is the intelligent Ventricular Assist Device (iVAD). The device functions as an artificial muscle wrap that assists the failing heart by applying compressive force — synchronous to its native rhythm — around the external surface of the heart's ventricles. The cyclic squeezing action supplements the heart muscle's efforts, leading to improved output from the diseased heart.
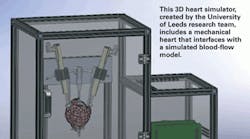
Researchers needed to physically apply the iVAD to the heart simulator to measure its compressive efforts, so a realistic in-vitro testing environment was imperative for its development. Traditional approaches for heart assist devices involve bulky mechanical mock circulatory systems or excised hearts that are metabolically supported by another animal's blood flow. Because the Leeds research team didn't want to use either method, they created a unique hardware-in-the-loop (HIL) heart simulator that combines a real-time software blood-flow model with a physical 3D mechanical heart. They also used the NI LabVIEW graphical programming environment and CompactRIO from National Instruments, Austin, to further enhance the testing environment so that the heart simulator could operate as a standalone system and run reliably for prolonged periods.
Heart simulator concept and design
HIL is a testing technique commonly used in industry, but why did the Leeds researchers choose it? With an assist device such as the iVAD, the interaction between the device itself and the heart surface is crucial — but depends on physical features that are difficult to model virtually, such as backlash and nonlinear friction. Therefore, it was imperative that the heart simulator include a physical object onto which the research team could apply the iVAD and monitor its raw compressive operation.
In addition, the Leeds engineers wanted a reconfigurable heart simulator, to physically and hemodynamically replicate different patient groups, illnesses, and animal models. Adaptability reduces the need for animal testing because it allows the heart simulator to be used for prolonged trials of prototype iVADs, as well as provide information on the iVAD's physiological effects.
In short, HIL simulates some components with software, and links these to physical pieces of hardware from the same system. For the heart simulator, a mechanical heart functions as hardware within a simulated blood-flow model loop. Then the continuous feedback loop between them is used to assess how the iVAD device's physical assistance affects the heart and blood flow if implanted inside the body.
The mechanical heart's shape is defined by two modifiable semicircular patterns of buckled spring steel strips, which are attached at both ends with adjustable boundary conditions. A custom-built NI vision program helps define the necessary boundary conditions to match the profile of each steel strip to a reference heart model. Two linear actuators cyclically flex the steel strips and realistically represent the dynamic movement of the heart's right and left ventricles. Actuator motion within the blood-flow model is controlled to mimic the simulated heart, so that the physical heart immediately reflects any volume change to the simulated heart.
In addition to matching the heart shape, the arrangement allows local variation of stiffness around the circumference of the mechanical heart by individually altering the mechanical properties of the strips, such as thickness. A thin elastic skin is used to surround the strips and apply the iVAD.
Simulator implementation
As mentioned, a feedback loop is used to assess the iVAD's assistance to the cardiovascular system. Four conformable pressure sensors are located at equal intervals around the mechanical heart to provide iVAD assistance (compression) data. Within the model, this data is converted to an assistive pressure for each ventricle; the subsequent effect on blood flow is calculated in real time and causes associated changes in the mechanical heart's motion.
The blood-flow model operates as a closed-loop lumped parameter model based on an electrical network analogy. Because each region of the heart is modeled separately, the heart is controlled locally to implement specific heart conditions and diseases. In addition, the blood-flow model can be automatically adjusted to represent real physiological data through the use of a nonlinear least-squares parameter estimation method implemented as a state within the LabVIEW code. This means that the heart simulator can accurately reflect the hemodynamics of most patient groups and in-vivo models, helping improve understanding of the device's potential effect.
CompactRIO controls the mechanical heart, runs simulations, and sends data to the Windows host for display and saving. The real-time controller executes two parallel loops — a high-priority control loop for the blood-flow model and a low-priority communication loop that sends and receives queued TCP data to and from the Windows host. The high priority blood-flow model loop runs at 500 Hz and converts the two ventricular volumes to a calibrated positional voltage that is sent to the FPGA I/O for each of the linear actuators to follow. The FPGA is compiled to handle all I/O from the CompactRIO as well as provide proportional-integral (PI) control of a heater, which is used to keep the heart simulator enclosure at a constant body temperature.
For more information, visit ni.com. This month's case study provided by Dr. David Keeling, School of Mechanical Engineering, University of Leeds, UK